Shenzhen Alu Rapid Prototype Precision Co., Ltd.
Industry News
- Home
- News
- How to medical device prototyping?
Prototyping a medical device is a rigorous process that balances functional innovation with strict regulatory compliance. Unlike consumer electronics, every iteration must be documented to ensure safety, efficacy, and "traceability" for future FDA or CE certification.
Here is the strategic workflow for medical device prototyping:
1. Requirement Definition (Design Inputs)
Before physical building begins, you must define the Design Inputs. This involves translatinguser needs (e.g. "must be easy to hold") into technical specifications (e.g., "handle diametermust be 30mm + 2mm with a high-friction TPE overmold").
Risk Analysis: Identify potential failure modes (ISO 14971) early.
Regulatory Path: Determine if the device is Class I, Il, or IIl, as this dictates the level ofdocumentation required during prototyping.
2. Low-Fidelity "Proof of Concept" (PoC)
The goal here is to test the core mechanism or "the math" behind the device. These models are often "looks-like/works-like" prototypes where the aesthetics are ignored to prove the function.
Materials: Cardboard, 3D-printed PLA, or off-the-shelf components.
Bench Testing: Use these to gather preliminary data. For example, if you are prototyping a fluid delivery system, you might use a basic pump and sensors to verify flow rates.
3. High-Fidelity Technical Prototyping
Once the PoC is verified, you move toward production-grade materials and processes. This stage is critical for human factors engineering (usability testing).
CNC Machining: Used for high-precision metal components (e.g., surgical Grade 5 Titanium or 316L Stainless Steel) or engineering plastics like PEEK and Delrin that require high biocompatibility and strength.
Injection Molding (Bridge Tooling): Using aluminum molds to create small batches (50–100 units) of parts in final materials like medical-grade PC-ASA or Silicone.
Electronics Integration: Moving from breadboards to custom PCBAs that fit within the device housing.
4. Design for Manufacturing (DFM)
A common pitfall is creating a prototype that cannot be mass-produced.
Draft Angles: Ensure all molded parts can be ejected from a tool.
Tolerances: Balance the high precision required for medical safety with the cost-effectiveness of manufacturing.
Assembly: Design the device to be assembled easily and correctly every time (Poka-yoke).
5. Verification and Validation (V&V)
This is the final hurdle before clinical trials or market submission.
Verification: "Did we build the device right?" (Testing against the technical specs).
Validation: "Did we build the right device?" (Testing against user needs through clinical evaluation).
Biocompatibility Testing (ISO 10993): Ensuring the materials do not cause adverse reactions when in contact with the human body.
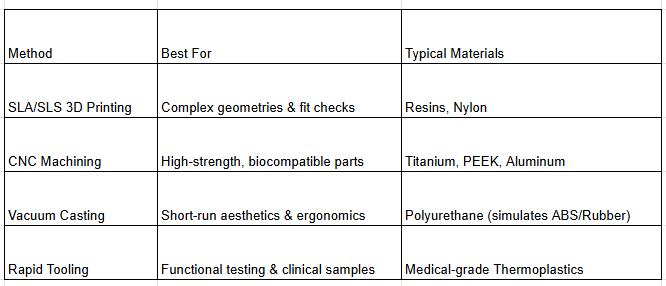
Comparison of Prototyping Methods